Calculating formal charge1/26/2024 Also, generally, the oxidation state of oxygen is -2 (but in peroxides and super-oxides it may vary).But sometimes it is -1 (when bound to alkali or alkali earth metals.) Further, the oxidation state of hydrogen is usually +1.Meanwhile, the oxidation state of fluorine is always -1.The oxidation state of alkali metals is always +1, and for alkali earth metals it is +2.The total charge of a molecule or ion is the sum of the charges of each atom.Oxidation state of a single element is zero (this includes the molecules made of a single element as well).The mathematical relationship for this calculation is as follows:įormal charge = (number of valence electrons in neutral atom)- (number of lone pair electrons) – (*bond pair electrons)įigure 02: Oxidation States of Atoms in Different Molecules Tips for the Determination of Oxidation State: 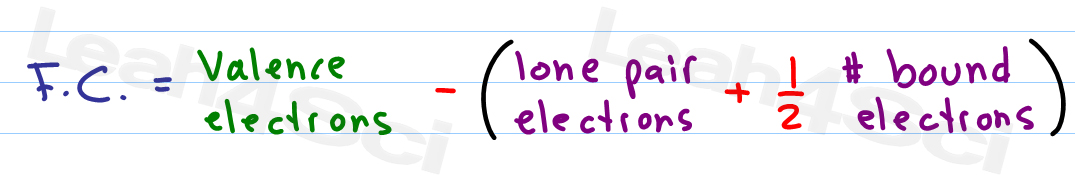
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
